|
Marianne Davies, DNP, ACNP, AOCNP, FAANAssociate Professor, Yale School of NursingYale University West Haven, CT Oncology Nurse Practitioner-Thoracic & Solid Tumor Senior Advanced Practice Provider II Smilow Cancer Hospital at Yale-New Haven Yale Comprehensive Cancer Center New Haven, CT |
 |
Shawn Perkins, RN, BSN, OCNThoracic Nurse NavigatorUNC Cancer Hospital Chapel Hill, NC |
Introduction
For many patients with non–small-cell lung cancer (NSCLC), immunotherapy is now becoming the standard of care. Immunotherapy given as monotherapy or in combination with other agents has contributed to promising results in NSCLC, with the earliest results observed in 2015 with nivolumab, and subsequently leading to FDA approval of 6 agents. These agents comprise a group of immune checkpoint inhibitors (ICIs) that act on programmed cell death protein 1 (PD-1; nivolumab, pembrolizumab, cemiplimab), programmed death-ligand 1 (PD-L1; atezolizumab, durvalumab), or cytotoxic T-lymphocyte–associated protein 4 (CTLA-4; ipilimumab).1 Long-term outcomes from phase 3 clinical trials suggest that immunotherapy contributes to improved lung cancer survival, making it a chronic, treatable disease for many patients.2 However, ICIs function by disrupting the homeostatic mechanisms involved in the regulation of immune-cell function, which maintains balanced immune responses such that they may contribute to a unique spectrum of immune-related adverse events (irAEs) in some patients.3,4
Clinicians have been encouraged to engage patients in a shared decision-making process regarding available treatment options.5,6 This patient-centered process involves providing information on diagnosis, prognosis, and available treatments to patients and caregivers.7 The shared decision-making process helps to facilitate patient preferences and choices in line with their personal goals, with the anticipation of providing optimal care.
Decision support interventions (DSIs), or decision aids, can help facilitate the treatment decision-making process.7 DSIs can take the form of videos, educational booklets, and online or computer-based applications to present information about decisions and available alternatives, and to help patients clarify their individual preferences.8,9 Use of DSIs has been associated with increased patient knowledge, decreased decisional conflicts, and increased patient satisfaction regarding their care.10 Furthermore, DSIs can enhance satisfaction with provider communication and decrease anxiety related to decision-making in patients with late-stage cancer.11
In this regard, patients should receive up-to-date and timely education on mechanisms of action underlying immunotherapy, the clinical outcomes to expect with immunotherapy, and potential irAE manifestations. This article reviews the importance of patient education in patients with advanced NSCLC treated with immunotherapy. In addition, expert perspectives on patient education will be provided by Marianne Davies, DNP, ACNP, AOCNP, FAAN, an associate professor at Yale University, New Haven, CT, and a thoracic oncology nurse practitioner at the Smilow Cancer Hospital, Yale Comprehensive Cancer Center; and Shawn Perkins, RN, BSN, OCN, a nurse navigator from UNC Cancer Hospital, Chapel Hill, NC, who has lung cancer navigation experience in a community setting.
Importance of Patient Education
The importance of patient education is supported by guidelines and in the literature and has changed from a one-size-fits-all approach to individualized education that is tailored based on a specific treatment.12-18
When explaining immunotherapy to patients, plain language can be used to improve understanding.19 For example, patients should be told that ICIs work with the immune system to fight cancer. This can result in unique response patterns and side effects that differ from the cancer treatments patients may have previously received (such as chemotherapy). Tumors can avoid immune system detection through upregulating pathways that limit immune responses.20,21 ICIs block these pathways, restoring balance to a patient’s immune response by allowing T-cells to work against cancer cells.21-23
“Initially, the physician will begin the education about the treatment recommendation and the rationale for why they selected or are making the recommendation prior to the start of the therapy,” explained Dr Davies, who has been taking care of patients with lung cancer as a nurse practitioner for 20 years. “They’ll provide an overview of the mechanism of action, the treatment schedule, the potential risks and benefits, but based on the clinical trial data. As a team, we have conversations together about our messaging so we’re all delivering the same message to patients. We share real world experience with immune-oncology therapy through different lenses, to improve patient understanding to provide consistent reinforcement to maximize patient understanding of the information.”
Patient education is also critical for maximizing early recognition and reporting of irAEs. Patients must receive up-to-date and timely education on how to recognize, mitigate, manage, and report irAE manifestations to the healthcare team. “Every patient, during their structured education session, gets printouts of the drug-specific information that includes adverse effects that they can experience,” explained Mr Perkins, who has been involved in every aspect of the lung cancer trajectory for more than 10 years in a community facility. “We do a lot of education verbally as well, talking about the side effects, the inflammation, and the specific AEs that are emergencies. We really want to know about all of them—even ones where a patient may be managing those at home and they’re okay, we want to know about it as soon as possible. As soon as the clinic’s open, we want them to leave a message with our triage line, let us know what’s going on so that we can follow up with that patient. If there needs to be an intervention, we definitely don’t want it to progress to be a serious side effect or event.”
Effective patient education on the optimal duration for monitoring irAEs is unknown, and appropriate educational tools and outcome measures do not exist. As a result, developing effective patient education and establishing standardized outcome measures are needed to ensure high-quality patient care. Results from an educational initiative focused on treatment decisions and management of side effects for nearly 9000 patients with NSCLC and their caregivers revealed an 8% improvement in increased comprehension that treatment-related side effects need to be reported while on therapy and following immunotherapy, a 16% improvement in the understanding of the mechanism of action for immunotherapy, and a 26% improvement in an increased recognition that response to immunotherapy may take longer than chemotherapy.24
In a retrospective study of 159 patients with advanced lung cancer, 10.7% of the patients required hospitalization due to severe irAEs, and 52.9% visited the hospital following telephone consults.25 These findings suggest that early detection of irAEs before they become severe may be possible through education.
Selection of Treatment
Prior studies have found that patients with incurable cancer often misperceive that chemotherapy treatment goals are to cure; however, how patients perceive the curative potential of immunotherapy is unclear.26 In a single-institution survey of patients who initiated ICI therapy for advanced melanoma or NSCLC, the authors identified knowledge deficits in their patients regarding the mechanism of action of immunotherapy and lack of awareness about the timing and severity of side effects.26 Additionally, 34% of participants reported that the primary treatment goal of ICI therapy is to cure their cancer. These findings highlight the need for educational patient tools to supplement information received from oncology teams.
“I think it’s important to take a step back and assess what the patient knows and what their level of understanding, their education level, their cognitive level are—all those things play a part,” Mr Perkins explained. “As far as helping them manage those expectations, I think it’s important that you assess what they think this medication is going to do. A lot of people think, ‘Oh, I’m going to get this drug, it’s going to cure me.’ And some medical oncologists are great at putting everything out there and then some hedge because they don’t want to have those difficult conversations very early on. They don’t want to get rid of hope and sometimes they aren’t as forthcoming with the patient as they should be.”
The increase in product-to-consumer advertising has led to patients requesting drugs with the expectation and hope that there will be no side effects and they will have great clinical responses and prolonged survival.18 Clinicians need to explain to patients when they are not eligible for immunotherapy and discuss the actual response rates and the potential for toxicities associated with ICI therapy.
“One of the first things that we are challenged with is that some patients come into the clinic and say, ‘I don’t ever want to get chemotherapy,’” said Dr Davies. “They may be requesting immunotherapy monotherapy, so the challenge is educating the patient so they understand why monotherapy might be selected versus a combination regimen based on that patient’s particular profile. Setting expectations of when you can expect to see a potential side effect and what to monitor for is different for immune therapy versus chemotherapy, and so they need to have that reinforced. And then the other expectation really is when you can expect to see a response from immunotherapy.”
Clinicians must also educate patients on the expected response to treatment, namely that ICIs can result in long-term responses and survival even if the initial response is delayed or if progression is experienced before a response to treatment is observed.19,27 Patients should be apprised that responses to immunotherapy can vary among patients and from typical response patterns seen with chemotherapy or radiotherapy because immune responses can occur months after treatment, even after an initial increase in tumor size or appearance of new tumors.21,28,29
When selecting treatment for individual patients, clinicians should inform patients regarding the available options, and there should be a shared decision-making process where specific agents are discussed. The treatment selection process often involves multidisciplinary teams that may include a pulmonologist, medical oncologist, radiation oncologist, thoracic surgeon, interventional pulmonologist, radiologist, interventional radiologist, pathologist, palliative care specialist, clinical nurse specialist/nurse support, team coordinator, psychologist, clinical trials coordinator, nutritionist, and physical/occupational therapist.30
Originally established to diminish gaps in cancer care among marginalized populations, cancer navigation programs are becoming more common in both the United States and Canada.31,32 These programs may include social workers, lay health workers, or nurses who have been trained as patient navigators to help patients achieve care goals and overcome modifiable barriers to care, providing an individualized approach for the needs of each patient.33-35 Navigator responsibilities can include coordination of care, education, removal of medical system barriers, insurance coverage assistance, assistance with financial barriers, and providing emotional support.31,36-40
“We don't use the term navigator in our clinic. We have an oncology care coordinator (OCN) who is a nurse that follows up with patients after they go home,” Dr Davies explained. “The OCN makes follow up phone calls, helps to facilitate access to additional support resources in the community if the patient needs some different help, and then then communicates that information back to treatment team. If patients hadn't already called in with potential new side effects, the OCN will discuss with the primary team to assess if patients need to come in for further follow up. If the patient had reached out to the team, the triage practice nurse will assess the need for further follow up evaluation. So, there's several different support structures in place to help support the patient through their treatment regimen.”
“Really it’s a team effort, the education,” explained Mr Perkins. “The medical oncologist does the high-level education. The patient doesn’t usually retain a lot of that, they’re very overwhelmed. And part of the job as the nurse navigator is sitting down with the patient after the physician visit and assessing what’s been under said and understood as well as what’s still left to be explained. I’d say the lion’s share of education becomes the responsibility of the infusion navigator and the nurse navigator.”
DSIs may help facilitate the treatment decision-making process when multiple therapeutic options are available.7 A novel DSI was developed to engage patients with late-stage NSCLC in shared decision-making regarding treatment.41 This DSI included care plan cards that answered common questions patients have regarding treatment options, including immunotherapy, and a companion tool for patient preference clarification to assist in the shared decision-making process. Pilot testing revealed use of the DSI was associated with increased patient awareness of treatment options and their associated risks and benefits, with a decrease in patient concern about treatment cost and uncertainty in decision-making.
“I have used various methods to help educate patients on immunotherapy,” Mr Perkins noted. “In my community setting, patients wanted something in hand, whether that’s a picture that I’ve drawn with a few notes along with my explanation of things, or whether that’s a printout or a brochure. In my academic setting in Chapel Hill, it seems that patients have more health literacy and are a lot more tech savvy and they’re fine with me sending them a MyChart message through our EMR that has links to educational videos or to visuals of how a biopsy’s going to be taken.”
Training materials developed for clinicians to help address shared decision-making and to improve patient-centered care resulted in increased participation in shared decision-making from 34% pre-education to 88% following education with training materials.42 Areas of particular improvement included a 58% improvement in patient understanding of reasonable treatment options and an 88% improvement in helping to determine patient goals and preferences.
“The first step really is assessing their health literacy and assessing what patients want to know and how much they want to know or how much their family members want to know,” Dr Davies said. “Some patients really don’t want a lot of information because it can increase their level of anxiety, so we assess what their need is and then provide material that is necessary in alignment with their requests.”
In the open-label, phase 3 Checkmate 816 study, where patients with stage IB to IIIA resectable NSCLC were randomized to receive platinum-based chemotherapy with or without nivolumab, followed by resection, those who received neoadjuvant nivolumab had significantly prolonged event-free survival, and more patients achieved pathological complete response compared with those who received chemotherapy alone.43 Therefore, there is a need to understand how patients feel about neoadjuvant immunotherapy and delaying surgery, and what the nurse/nurse navigator can do to educate the patient about neoadjuvant therapy.
“There’s a lot of anxiety and fear there,” Mr Perkins explained. “I think patients are going to have anxiety [about neoadjuvant immunotherapy] and you really have to try to dispel myths and talk about the benefit versus the risk. Talking through the benefits seems to be more helpful to my patients than anything. But I think that is an important thing to mention, that [neoadjuvant therapy with nivolumab plus chemotherapy] doesn’t complicate the surgery, it doesn’t change how successful the surgery’s going to be in any way.”
“If a patient meets treatment criteria, meaning that they do not have other comorbid medical reasons, why they should not get neoadjuvant therapy with immune therapy, then they become a candidate that you can have that discussion with,” explained Dr Davies. “The clinical data do support improved long-term outcomes for patients that do receive neoadjuvant nivolumab therapy. If you can have an improved surgical outcome and long-term outcome, then that would be our recommendation.”
When asked about patient concerns and anxiety associated with delaying surgery to receive systemic neoadjuvant immunotherapy, Dr Davies noted, “A lot of the reasons that they might be reluctant is because they’re so anxious because they’ve just been diagnosed. There are many fears associated with that—a fear of the diagnosis and what are the outcomes and the unknowns."
“We have patient support groups that meet, and our licensed clinical social worker will meet with all the patients, particularly when they’re newly diagnosed, and then ongoing throughout the treatment to help allay some of those concerns and help provide management strategies for dealing with stressors.”
Preparing Patients for irAEs
Immunotherapy poses risks of irAEs that require prompt recognition in order to minimize morbidity and mortality.26 Although serious irAEs are infrequent and treatable, patient education is very important. Teaching should begin prior to the decision to initiate immunotherapy and include both patients and their caregivers because early recognition of irAEs is essential to effective treatment. And, unlike chemotherapy, irAEs tend to be delayed.44 Patients should be reminded that irAEs are different from traditional chemotherapy side effects.18 Side effects of ICIs can occur any time, but most occur within 3 months of starting treatment and can develop even up to 1 year after ending treatment.19
“One of the key things is for patients and family members to have a really good understanding of what a patient’s baseline symptoms are,” explained Dr Davies. “We incorporate simple terms to assess changes, such as ‘If you used to be able to walk up a flight of stairs and now you have trouble walking 10 yards and you get more winded, that’s a significant change. That would be something you need to call us about.’ I say, ‘This is why I’m asking all these questions, it’s because these side effects can occur at any time, even months after treatment, even after you’ve completed therapy and maybe moved on to something else.’ My job is to do that assessment at each visit and reinforce the fact that we need to know of any new symptoms and do our due diligence in terms of monitoring for laboratory abnormalities and abnormalities found on diagnostic imaging.”
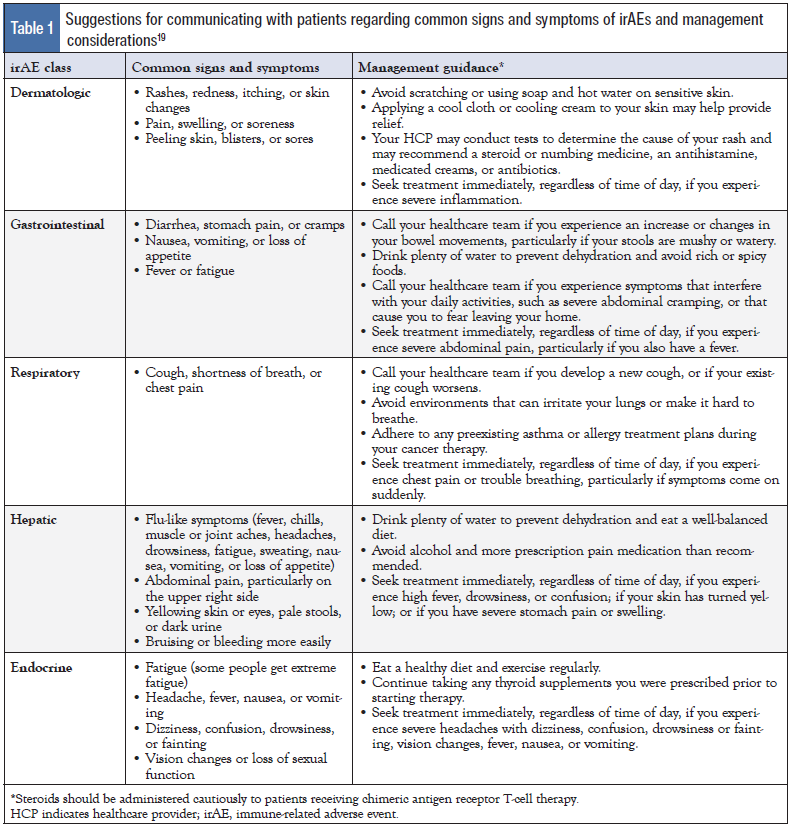
Patients should be instructed to immediately report the following: increased shortness of breath or nagging frequent cough; significant diarrhea associated with abdominal pain, cramping, or blood/mucus in the stool; heart palpitations; severe malaise; significant rash with hives or significant itching with rash; and any other symptoms that seem concerning to the patient.18 Serious irAEs may require treatment discontinuation; however, prompt recognition and management may lead to resolution of most irAEs, and patients can often continue treatment.12,15,45,46 As shown in Table 1, language related to common signs and symptoms of irAEs and management should be tailored differently for patients than for clinicians.
Patients should also be educated on when they may be able to manage irAEs themselves and when they should reach out to their healthcare team.19 Patients should alert the cancer care team and other healthcare professionals (HCPs) if irAEs are experienced or if any symptoms change or worsen. Patients should stay in contact with their cancer team and alert other HCPs that they are receiving or have received ICIs.
“Educating their family members is important because that wife may be the only one that knows that he’s been in the bathroom more times than he should be and he’s not going to say anything,” Mr Perkins explained. “And I think it’s just constant reinforcement, constant encouraging. We need to know—it’s important that we know. It doesn’t mean that you’re going to stop treatment. That could be one option, but that’s not the only option. And I think that our patients know that if you tell me what’s going on earlier, we have the best chance of taking care of it efficiently and preventing you from stopping treatment. And when you frame it that way, I think you get a little bit better compliance, too.”
Financial Toxicity
Financial distress related to cancer has been linked to an increased risk of psychosocial distress and poorer quality of life. In a study of 120 insured, affluent, and well-educated patients, 29% had “high or overwhelming” financial distress, 65% had clinically significant levels of overall distress, and 66% reported a component of emotional distress.47 Furthermore, among 3724 patients with cancer, 52% reported high levels of psychological distress, which indicates that these patients are inherently at greater risk for psychosocial distress without the added burden of financial stressors.48 Financial burden has also been linked with poorer quality of life. Patients make many adjustments in response to their cancer, and financial stress may negatively impact overall well-being.
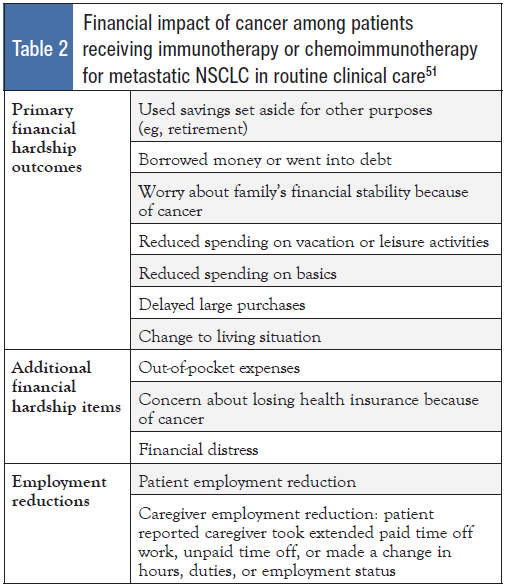
In the 2010 National Health Interview Survey, patients reporting “a lot” of financial problems were 4 times less likely to report “good” or better quality of life compared with those without financial problems.49 This link between financial toxicity and poorer quality of life has been demonstrated in different cancer types, including lung cancer.50 In a survey of 60 patients with metastatic NSCLC receiving immunotherapy or chemoimmunotherapy, financial hardship was common, with 33% reporting material, 63% reporting psychological, and 57% reporting behavioral hardship.51 Variables related to financial impact are shown in Table 2.
“It’s not just about the cost of the drug either,” said Dr Davies. “Depending on the frequency with which they’re coming into the clinic, the financial toxicity can include lost time from work for the patient or their family member caregiver, the cost of transportation, the price of gas; can the patients afford to keep coming back and forth? Do they have to pay for parking? Do they even have a car? Do we have to arrange for transportation? That’s when we really have to work with the patient and talk about that impact on their finances, because we all know that in patients that are suffering from financial toxicity, there’s decreased adherence to medication regimens if they can’t afford them, so we need to have that discussion.”
Providers have a responsibility to deliver high-quality care for patients while practicing good resource stewardship. In 2015, the American Society of Clinical Oncology (ASCO) published a framework to provide a physician-guided tool for comparing new therapeutic options with the current standard of care for advanced cancer and curative treatment.52 Notably, ASCO highlighted that discussions regarding cost are a critical component of high-quality care for patients.53 Although concerns related to cost are prevalent among cancer patients, they are not frequently addressed. In a survey of 132 patients, 47% reported high levels of financial stress; however, 71% reported that they rarely had discussions with their providers related to cost.54
Engaging patients in cost discussions has been shown to contribute to reduced out-of-pocket expenses. In a study of 300 patients, 19% reported speaking with their physician regarding cost, with 57% reporting reduced financial burden following the discussion.55 Failure to discuss out-of-pocket costs may be associated with medication nonadherence.56 Patients who are most likely to engage in cost discussions are thought to be those with the greatest risk of experiencing significant financial burden, and subsequently those who have increased risk for noncompliance to treatment. Screening for patients who may be at risk for financial toxicity should be implemented as part of regular clinical practice due to the consequences of high out-of-pocket costs. One potential tool is the COmprehensive Score for financial Toxicity measure, which is a validated patient-reported outcome measure to assess for financial toxicity in patients with cancer.57
“In the community setting, which is my experience with our lung cancer patients, we didn’t have financial navigators,” Mr Perkins said. “I think every patient needs financial navigation and that should be one of their first stops because that’s also a huge source of anxiety and worry, and it impacts treatment decisions. Patients decide not to get treatment because of what it’s going to cost, especially if they realize that it’s not likely to result in a cure. They start weighing, ‘Well, how much is my time worth? And how much burden am I going to leave my family?’ So that’s a big need.”
Patients may benefit from education regarding cost from discussions with their providers and from available resources. Most oncologists agree that these discussions comprise a critical component of shared decision-making; however, a large proportion express discomfort with these discussions.58 Some oncologists believe that the clinical setting is not an appropriate place to discuss cost, which should not play a role in clinical decision-making. However, affordability conversations help patients better cope with the financial concerns around optimal care. ASCO highlighted that providers have a responsibility to engage patients in these discussions to help address the rising cost of cancer care.
Conclusion
For many patients with NSCLC, immunotherapy is now becoming the standard of care. Patients must receive up-to-date and timely education on mechanisms of action underlying immunotherapy, factors in selecting therapy, the clinical outcomes that can be expected with immunotherapy, and recognition and management of potential irAE manifestations. The treatment selection process often involves multidisciplinary teams that may include members of various specialties, including medical oncologists, advanced practice oncology nurses, and nurse and patient navigators. Indeed, navigation programs are becoming more prevalent to assist with care goal achievement and overcoming modifiable barriers to care. Nurse navigators can also help with discussions around medication cost, which is also a critical component of high-quality patient care and may help patients cope with care-related financial concerns.
“I think there’s a lot of education that could be done that isn’t because in healthcare we get in a hurry with everything we do,” Mr Perkins said. “And sometimes it’s easy to just give a high-level explanation that really requires the patient to understand how immunotherapy works and what side effects they’re likely to see. A lot of our patients are like my parents. And if somebody says they should get this treatment, they’re going to find somebody they know that’s had it, or somebody that’s heard of somebody that’s had it, and ask them how that went. And nobody likes to tell the good stories. Everybody likes to get the shock value of the not-so-good stories.”
“One of the most important things that we need, as healthcare providers, as nurses, as practitioners across our team, is to keep up to date with the latest guidelines because so much of the management and work-up, the evaluation of these toxicities, was based on our knowledge of other autoimmune diseases and their management,” said Dr Davies. “And now we actually have studies that are looking at appropriate management, so it’s important as healthcare providers that we keep up to date with the latest guidelines. It’s important that we incorporate those care plans into our practice and know what the newest information is so that we can best educate our patients and their caregivers.”
References
- Rodak O, Peris-Diaz MD, Olbromski M, et al. Current landscape of non-small cell lung cancer: epidemiology, histological classification, targeted therapies, and immunotherapy. Cancers. 2021;13:4705.
- Reck M, Ciuleanu TE, Cobo M, et al. First-line nivolumab plus ipilimumab with two cycles of chemotherapy versus chemotherapy alone (four cycles) in advanced non-small-cell lung cancer: CheckMate 9LA 2-year update. ESMO Open. 2021;6:100273.
- Martins F, Sofiya L, Sykiotis GP, et al. Adverse effects of immune-checkpoint inhibitors: epidemiology, management and surveillance. Nat Rev Clin Oncol. 2019;16:563-580.
- Postow MA, Sidlow R, Hellmann MD. Immune-related adverse events associated with immune checkpoint blockade. N Engl J Med. 2018;378:158-168.
- Levinson W, Kao A, Kuby A, Thisted RA. Not all patients want to participate in decision making. A national study of public preferences. J Gen Intern Med. 2005;20:531-535.
- Leighl NB, Shepherd FA, Zawisza D, et al. Enhancing treatment decision-making: pilot study of a treatment decision aid in stage IV non-small cell lung cancer. Br J Cancer. 2008;98:1769-1773.
- Elwyn G, Edwards A, Hood K, et al. Achieving involvement: process outcomes from a cluster randomized trial of shared decision making skill development and use of risk communication aids in general practice. Fam Pract. 2004;21:337-346.
- Hopmans W, Damman OC, Senan S, et al. A patient perspective on shared decision making in stage I non-small cell lung cancer: a mixed methods study. BMC Cancer. 2015;15:959.
- Grossman Liu L, Ancker JS, Masterson Creber RM. Improving patient engagement through patient decision support. Am J Prev Med. 2021;60:438-441.
- Spiegle G, Al-Sukhni E, Schmocker S, et al. Patient decision aids for cancer treatment: are there any alternatives? Cancer. 2013;119:189-200.
- Flocke SA, Antognoli E, Daly BJ, et al. The role of oncology nurses in discussing clinical trials. Oncol Nurs Forum. 2017;44:547-552.
- Brahmer JR, Lacchetti C, Schneider BJ, et al. Management of immune-related adverse events in patients treated with immune checkpoint inhibitor therapy: American Society of Clinical Oncology Clinical Practice Guideline. J Clin Oncol. 2018;36:1714-1768.
- Wood LS. Immune-related adverse events from immunotherapy: incorporating care step pathways to improve management across tumor types. J Adv Pract Oncol. 2019;10(suppl 1):47-62.
- Davies MJ. PD-1/PD-L1 inhibitors for non-small cell lung cancer: incorporating care step pathways for effective side-effect management. J Adv Pract Oncol. 2019;10(suppl 1):21-35.
- Haanen J, Carbonnel F, Robert C, et al. Management of toxicities from immunotherapy: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2017;28(suppl 4):iv119-iv142.
- Schneider BJ, Naidoo J, Santomasso BD, et al. Management of immune-related adverse events in patients treated with immune checkpoint inhibitor therapy: ASCO Guideline update. J Clin Oncol. 2021;39:4073-4126.
- Naing A, Hajjar J, Gulley JL, et al. Strategies for improving the management of immune-related adverse events. J Immunother Cancer. 2020;8:e001754.
- Sandy B. Explaining immunotherapy to our patients with NSCLC. Medicine Matters Oncology. 2018. May 2, 2018. https://oncology.medicinematters.com/immunotherapy/toxicities/explaining-immunotherapy-to-our-patients-with-non-small-cell-lun/15345396. Accessed December 23, 2022.
- Wood LS, Moldawer NP, Lewis C. Immune checkpoint inhibitor therapy: key principles when educating patients. Clin J Oncol Nurs. 2019;23:271-280.
- Beatty GL, Gladney WL. Immune escape mechanisms as a guide for cancer immunotherapy. Clin Cancer Res. 2015;21:687-692.
- Kannan R, Madden K, Andrews S. Primer on immuno-oncology and immune response. Clin J Oncol Nurs. 2014;18:311-317, 326.
- de Mello RA, Veloso AF, Esrom Catarina P, et al. Potential role of immunotherapy in advanced non-small-cell lung cancer. Onco Targets Ther. 2017;10:21-30.
- Dougan M, Dranoff G. The immune response to tumors. Curr Protoc Immunol. 2009;Chapter 20:Unit 20 11.
- Herrmann T, Hamarstrom E, Carey C. Online patient education in advanced lung cancer: effect on patient/caregiver knowledge. J Thorac Oncol. 2017;12(1 suppl):S1110.
- Aso S, Kawamura N, Yanagida H, et al. Exploring the educational needs for severe immune-related adverse events of PD-1/PD-L1 inhibitors in advanced lung cancer: a single-center observational study. Asia Pac J Oncol Nurs. 2022;9:100076.
- Petrillo LA, Zhou AZ, Sullivan R, et al. Knowledge about risks, benefits, and curative potential of immunotherapy among patients with advanced cancer. Oncologist. 2021;26:e2090-e2093.
- Michot JM, Bigenwald C, Champiat S, et al. Immune-related adverse events with immune checkpoint blockade: a comprehensive review. Euro J Can. 2016;54:139-148.
- Fay AP, Brandao Moreira R, Nunes Filho PRS, et al. The management of immune-related adverse events associated with immune checkpoint blockade. Expert Rev Qual Life Canc Care. 2016;1. www.tandfonline.com/doi/full/10.1080/23809000.2016.1142827. Accessed December 23, 2022.
- Wolchok JD, Hoos A, O'Day S, et al. Guidelines for the evaluation of immune therapy activity in solid tumors: immune-related response criteria. Clin Can Res. 2009;15:7412-7420.
- Hardavella G, Frille A, Theochari C, et al. Multidisciplinary care models for patients with lung cancer. Breathe (Sheff). 2020;16:200076.
- Freeman HP. The history, principles, and future of patient navigation: commentary. Semin Oncol Nurs. 2013;29:72-75.
- Walkinshaw E. Patient navigators becoming the norm in Canada. CMAJ. 2011;183:E1109-E1110.
- Parker VA, Lemak CH. Navigating patient navigation: crossing health services research and clinical boundaries. Adv Health Care Manag. 2011;11:149-183.
- Pedersen A, Hack TF. Pilots of oncology health care: a concept analysis of the patient navigator role. Oncol Nurs Forum. 2010;37:55-60.
- Wells KJ, Battaglia TA, Dudley DJ, et al. Patient navigation: state of the art or is it science? Cancer. 2008;113:1999-2010.
- Fischer SM, Sauaia A, Kutner JS. Patient navigation: a culturally competent strategy to address disparities in palliative care. J Palliat Med. 2007;10:1023-1028.
- Shlay JC, Barber B, Mickiewicz T, et al. Reducing cardiovascular disease risk using patient navigators, Denver, Colorado, 2007-2009. Prev Chronic Dis. 2011;8:A143.
- Goff SL, Pekow PS, White KO, et al. IDEAS for a healthy baby--reducing disparities in use of publicly reported quality data: study protocol for a randomized controlled trial. Trials. 2013;14:244.
- Scott LB, Gravely S, Sexton TR, et al. Examining the effect of a patient navigation intervention on outpatient cardiac rehabilitation awareness and enrollment. J Cardiopulm Rehabil Prev. 2013;33:281-291.
- Darnell JS. Navigators and assisters: two case management roles for social workers in the Affordable Care Act. Health Soc Work. 2013;38:123-126.
- Myers RE, Advani SM, Myers P, et al. Engaging patients with late-stage non-small cell lung cancer in shared decision making about treatment. J Pers Med. 2021;11:998.
- Adelson KB, Herbst RS, Peterson P, et al. NSCLC: integrating the “Yale model shared decision-making solution” into the practice setting. J Clin Oncol. 2020;38(suppl):7054.
- Forde PM, Spicer J, Lu S, et al. Neoadjuvant nivolumab plus chemotherapy in resectable lung cancer. N Engl J Med. 2022;386:1973-1985.
- Green LM. Immunotherapy in cancer care: educating patients about what to expect. Oncology Nursing News. 2015;9. www.oncnursingnews.com/view/immunotherapy-in-cancer-care-educating-patients-about-what-to-expect. Accessed December 23, 2022.
- Puzanov I, Diab A, Abdallah K, et al. Managing toxicities associated with immune checkpoint inhibitors: consensus recommendations from the Society for Immunotherapy of Cancer (SITC) Toxicity Management Working Group. J Immunother Cancer. 2017;5:95.
- Weber JS, Postow M, Lao CD, Schadendorf D. Management of adverse events following treatment with anti-programmed death-1 agents. Oncologist. 2016;21:1230-1240.
- Meeker CR, Geynisman DM, Egleston BL, et al. Relationships among financial distress, emotional distress, and overall distress in insured patients with cancer. J Oncol Pract. 2016;12:e755-764.
- Mehnert A, Hartung TJ, Friedrich M, et al. One in two cancer patients is significantly distressed: prevalence and indicators of distress. Psychooncology. 2018;27:75-82.
- Fenn KM, Evans SB, McCorkle R, et al. Impact of financial burden of cancer on survivors' quality of life. J Oncol Pract. 2014;10:332-338.
- Lathan CS, Cronin A, Tucker-Seeley R, et al. Association of financial strain with symptom burden and quality of life for patients with lung or colorectal cancer. J Clin Oncol. 2016;34:1732-1740.
- McLouth LE, Nightingale CL, Levine BJ, et al. Unmet care needs and financial hardship in patients with metastatic non-small-cell lung cancer on immunotherapy or chemoimmunotherapy in clinical practice. JCO Oncol Pract. 2021;17:e1110-e1119.
- Schnipper LE, Davidson NE, Wollins DS, et al. American Society of Clinical Oncology Statement: a conceptual framework to assess the value of cancer treatment options. J Clin Oncol. 2015;33:2563-2577.
- Meropol NJ, Schrag D, Smith TJ, et al. American Society of Clinical Oncology guidance statement: the cost of cancer care. J Clin Oncol. 2009;27:3868-3874.
- Meisenberg BR, Varner A, Ellis E, et al. Patient attitudes regarding the cost of illness in cancer care. Oncologist. 2015;20:1199-1204.
- Zafar SY. Financial toxicity of cancer care: it's time to intervene. J Natl Cancer Inst. 2016;108:dvj370.
- Bestvina CM, Zullig LL, Rushing C, et al. Patient-oncologist cost communication, financial distress, and medication adherence. J Oncol Pract. 2014;10:162-167.
- de Souza JA, Yap BJ, Wroblewski K, et al. Measuring financial toxicity as a clinically relevant patient-reported outcome: the validation of the COmprehensive Score for financial Toxicity (COST). Cancer. 2017;123:476-484.
- Schrag D, Hanger M. Medical oncologists' views on communicating with patients about chemotherapy costs: a pilot survey. J Clin Oncol. 2007;25:233-237.