INTRODUCTION
In the United States, approximately 20,000 new cases of myelodysplastic syndrome (MDS) will be diagnosed each year.1 MDS has an increased prevalence with increasing age and affects males more frequently than females.1,2 Patients with MDS can be stratified into 2 broad subgroups: lower- and higher-risk disease.1,2 For patients with lower-risk MDS (LR-MDS), therapy is based on transfusion needs.2 The use of erythroid-stimulating agents (ESAs) is common, with a number of ESAs available for use.2
“Most patients with lower-risk MDS have a problem with anemia, with around half of patients becoming transfusion dependent. The current standard of care for patients with MDS is erythropoiesis-stimulating agents, which work around 30%-40% of the time and they generally stop working after 12 to 18 months. We have a significant unmet need for drugs which work after ESAs for a longer duration, and which leads to a higher rate of transfusion independence.”
- Dr Amer Zeidan, MBBS, MHS
Associate Professor, Yale University
Luspatercept modulates TGF-β signaling in MDS, which improves erythropoiesis.2 The MEDALIST trial (NCT02631070) was a multicenter, randomized trial of luspatercept for patients with red blood cell–dependent refractory anemia with ring sideroblasts who were not benefiting from the use of ESAs or were unlikely to benefit from use.2 In the MEDALIST trial, 37.9% of patients treated with luspatercept achieved transfusion independence compared to 13.2% of patients in the control group.2 Based upon the data from the MEDALIST trial, the US Food and Drug Administration (FDA) approved luspatercept in 2020 for treating anemia after failed treatment with an ESA.2 The COMMANDS clinical trial (NCT03682536) was designed to compare the use of luspatercept versus an ESA in transfusion-dependent patients with LR-MDS.2,3
The COMMANDS trial was a phase 3, open-label, randomized trial including 356 patients age ≥18 years with a diagnosis of MDS of very low risk, low risk, or intermediate risk requiring red blood cell transfusions of 2 to 6 packed red blood cell units per 8 weeks for ≥8 weeks immediately prior to study randomization.3 The median age of participants was 74 years, and 56% of patients were male.3 Patients were assigned in a 1:1 manner to receive luspatercept or epoetin alfa stratified by baseline red blood cell transfusion burden, ring sideroblast status, and endogenous serum erythropoietin concentration.3 The clinical trial evaluated the following treatment regimens: luspatercept subcutaneously once every 3 weeks starting at 1.0 mg/kg body weight with titration up to 1.75 mg/kg and epoetin alfa subcutaneously 450 IU/kg body weight with titration up to 1050 IU/kg with a maximum dose of 80,000 IU.3 The study primary end point was at least 12 weeks of red blood cell independence along with a mean hemoglobin increase of at least 1.5 g/dL.3 Safety was also assessed.3
Key Points3:
- The primary end point was reached for 59% of the luspatercept group and 31% of the epoetin alfa group (P<.0001). Patients who received luspatercept had a longer median treatment exposure (42 weeks) versus patients receiving epoetin alfa (27 weeks)
- Compared to epoetin alfa, luspatercept produced a more durable response of median transfusion independence (77 days vs 127 days, respectively)
- Luspatercept showed greater efficacy than epoetin alfa across various subgroups with LR-MDS
- Luspatercept was shown to have a manageable and predictable safety profile, consistent with the known profile in the approved indication
Currently, luspatercept is FDA approved to treat anemia in adult patients with beta thalassemia who require blood transfusions.4 It is also approved for adult patients with very low- to intermediate-risk MDS with ring sideroblasts or with myelodysplastic/myeloproliferative neoplasm with ring sideroblasts and thrombocytosis to treat anemia after an ESA requires ≥2 red blood cell units over 8 weeks.4
On May 1, 2023, the drug manufacturer’s supplemental New Drug Application for luspatercept as first-line treatment for patients with LR-MDS was accepted with a target action date of August 23, 2023.4
INTERVIEW WITH DR ZEIDAN
(conducted prior to the recent FDA approval of luspatercept for LR-MDS-associated anemia)
My name is Amer Zeidan. I am an associate professor of medicine at Yale University, and it is a true pleasure to discuss the COMMANDS clinical trial, which evaluated luspatercept against erythropoietin-stimulating agents (ESAs) in patients with lower-risk MDS, who are transfusion dependent for red blood cells. This study demonstrated that luspatercept is more effective with higher efficacy for transfusion independence and longer durability of transfusion independence than ESAs. We will be discussing the efficacy, the subgroup analysis, the safety profile, and the next steps with this drug and how it could impact the landscape of management of patients with LR-MDS.
Q: The COMMANDS clinical trial evaluated the use of luspatercept versus erythropoietin (EPO) alfa in patients with lower-risk MDS who are ESA naïve and transfusion dependent. In your opinion, what is the current unmet need in this patient group?
A: The COMMANDS study evaluated the efficacy and safety of luspatercept versus EPO alfa for the treatment of anemia due to lower-risk MDS with or without ring sideroblasts in patients who were 18 years of age or older. These patients had <5% blasts in the bone marrow, had endogenous serum EPO (sEPO) <500 U/L, required red blood cell (RBC) transfusions, and were ESA naïve (Figure 1). In my clinical experience, >90% of patients with LR-MDS have anemia and around half of these patients become RBC transfusion dependent. This is associated with significant complications and a higher death rate. Correcting the anemia is very important in patients with MDS, especially patients with lower respiratory disease. The current standard of care for the treatment of anemia due to lower-risk MDS is ESAs first line, but they only work about 30% to 40% of the time. In patients who have a response to ESAs they generally stop working after 12 to 18 months of use. This primary and secondary failure is a significant unmet need in these patients.
The COMMANDS trial rationale was to evaluate luspatercept, a drug that is already approved for use after ESA failure, as a first-line treatment and compare it with ESA to determine if it increases the rate or proportion of patients who achieve transfusion independence along with prolonging the duration of transfusion independence regardless of the presence of ring sideroblasts.
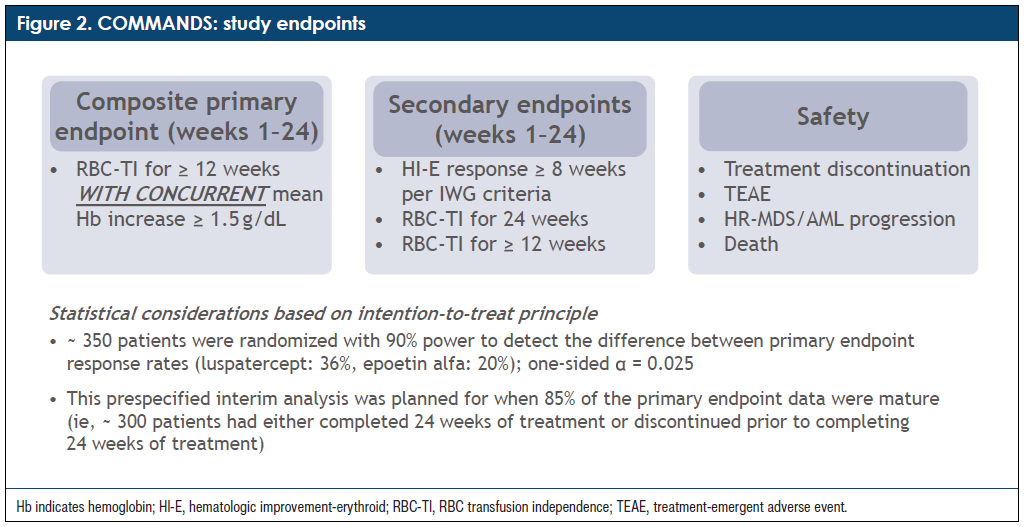
Q: The COMMANDS clinical trial had a composite primary end point of RBC transfusion independence for ≥12 weeks with a concurrent mean hemoglobin increase of ≥1.5 g/dL (Figure 2). The secondary end points were hematologic improvement – erythroid (HI-E) response ≥8 weeks per International Working Group (IWG) criteria, RBC transfusion independence for 24 weeks, and RBC transfusion independence for ≥12 weeks (Figure 2). How does the primary end point used in the COMMANDS trial differ from the similarly designed clinical trial for LR-MDS–associated anemia?
A: The primary end point of the COMMANDS trial was 12-week transfusion independence coupled with an average hemoglobin increase of 1.5 g/dL or higher for the 356 patients who went into the study (Figure 2). Classically, the end point in many of the studies that evaluated patients with LR-MDS with transfusion dependence focused on 8-week transfusion independence. We are realizing that to make a study more clinically relevant for patients, we need a longer duration of transition and dependence as transfusions can also be dependent on patient and physician preference, not only hemoglobin level. I think the 12-week transfusion independence and an objective hemoglobin rise are important as an improved end point over the traditional primary end point that has been used in LR-MDS clinical trials.
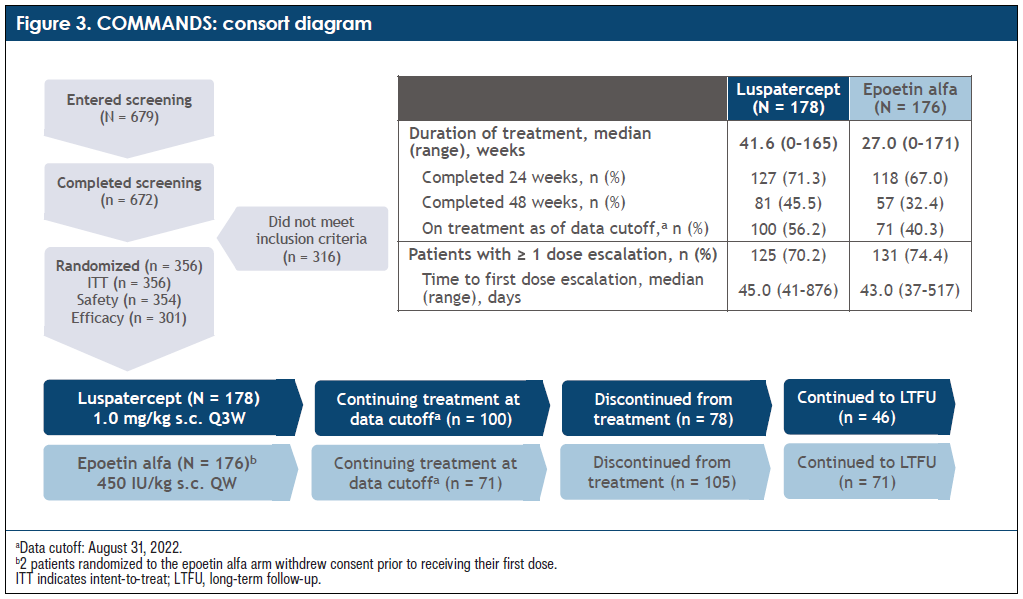
Q: In the COMMANDS trial, luspatercept was given to 178 patients at 1.0 mg/kg subcutaneously every 3 weeks with possible titration up to 1.75 mg/kg. Epoetin alfa was dosed at 450 IU/kg subcutaneously once a week with possible titration up to 1050 IU/kg (Figure 3). How did treatment with luspatercept compare to ESA standard of care in patients with LR-MDS–associated anemia in this study?
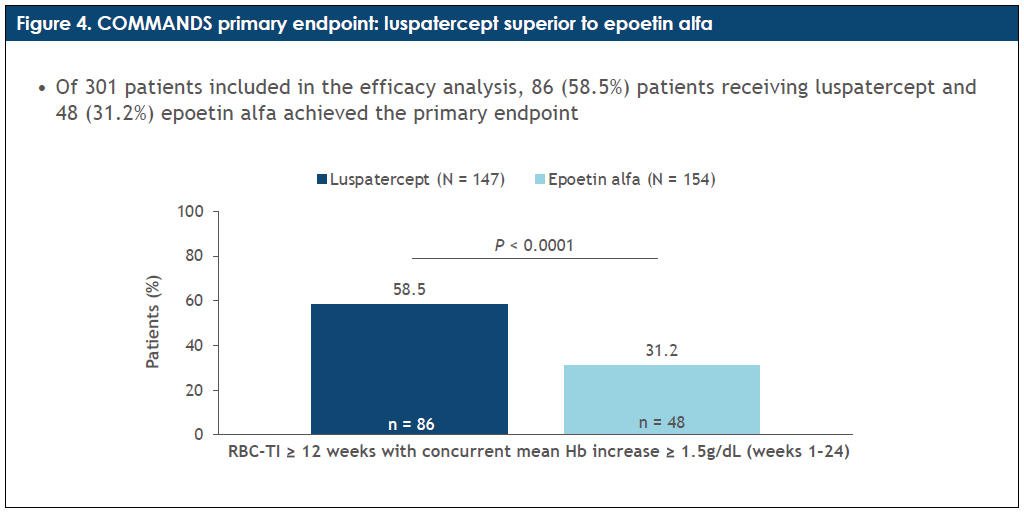
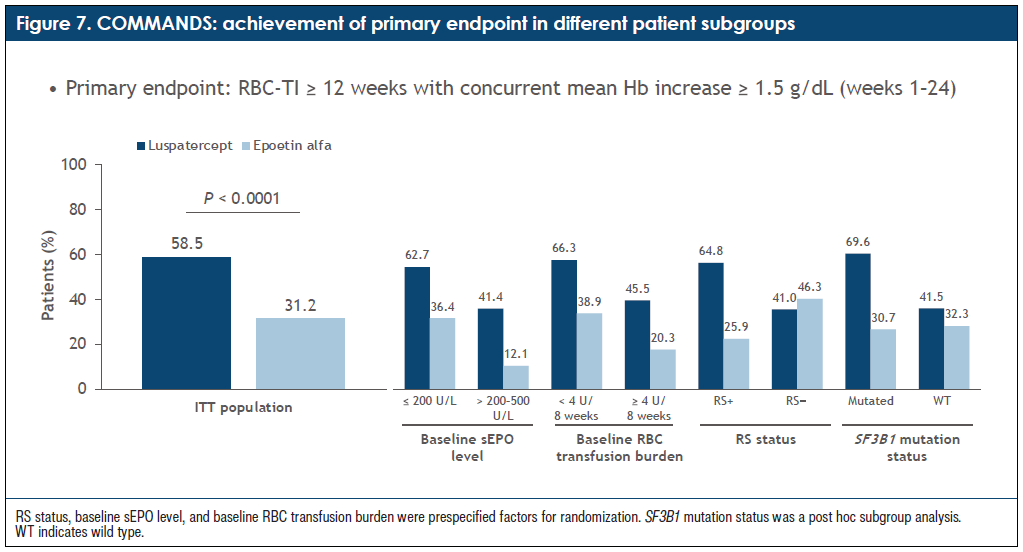
A: As previously discussed, the primary study end point is comparing luspatercept with short-acting ESAs. The study demonstrated that luspatercept increases the rate of transfusion independence at 12 weeks coupled with an increased hemoglobin rise of 1.5 g/dL for 58.5% of patients compared with 31.2% of patients receiving epoetin alfa (P<.0001) (Figure 4). This is a significantly higher rate, almost double the rate of transfusion independence with a longer duration, with the use of luspatercept.
Q: Data from the COMMANDS trial demonstrated that luspatercept had an advantage over epoetin alfa in treatment duration. What is your opinion on the durability of treatment in this study?
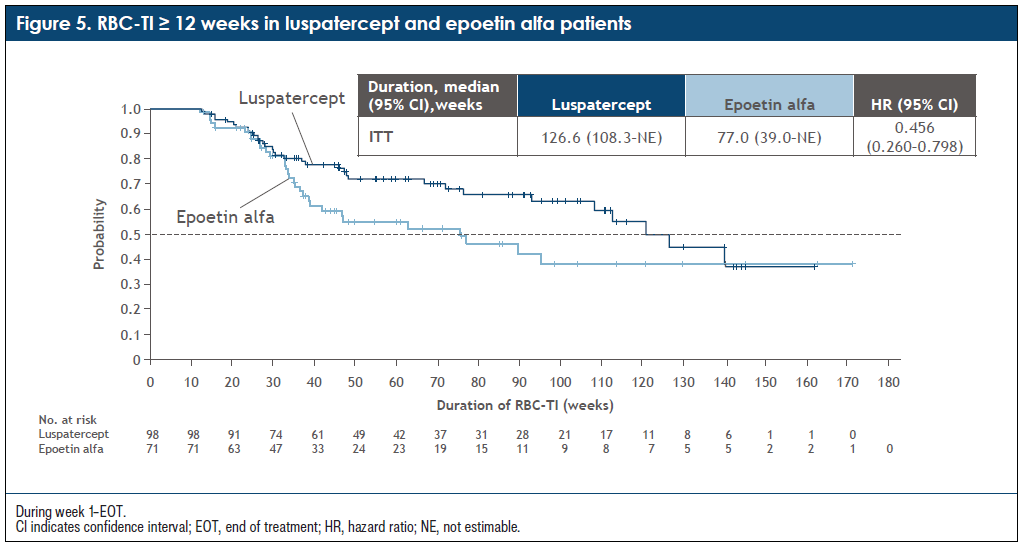
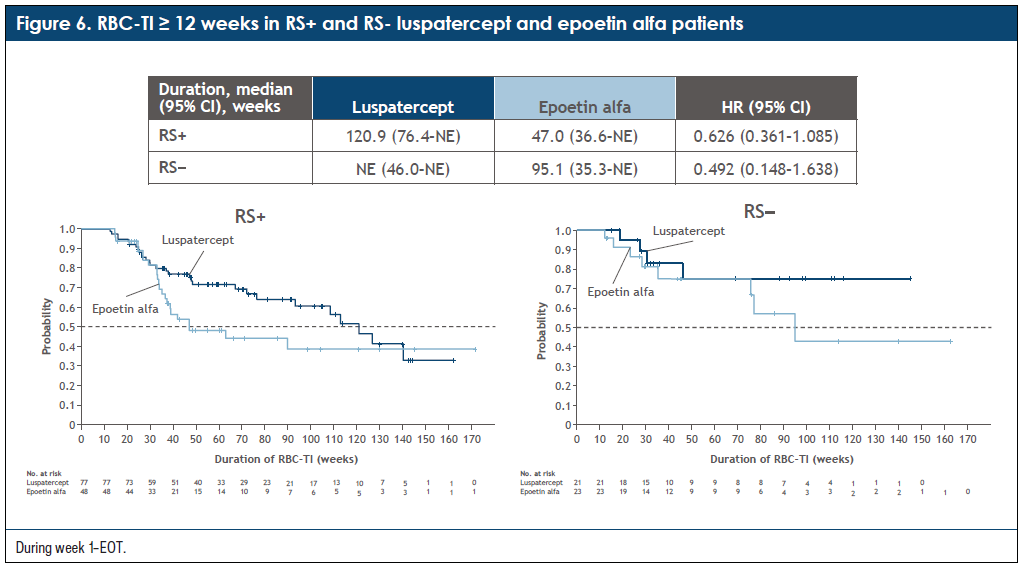
A: Some of my patients with LR-MDS live multiple years, and being transfusion independent has a significant impact on their quality of life and with complications. Having a well-tolerated durable treatment is important because ultimately these patients will require lifelong treatment. When treating patients with LR-MDS, we think about therapy sequencing and how we can optimize how long patients can stay on a therapy that is well tolerated and while they are not needing transfusions. Luspatercept has a median duration of transfusion independence of 2.5 years compared with 1.5 years with ESA, which is significant with some patients remaining even longer than that with transfusion independence (Figure 5 and Figure 6). I believe this duration is very important, as patients generally go from one treatment to the next.
Q: How do the study end points translate into benefit for patients with LR-MDS, and what are the most relevant benefits for patients with luspatercept?
A: In the subgroup analysis, luspatercept did well across different groups (Figure 7). For example, the study required patients to receive 2 to 6 units of blood every 8 weeks. Among the patients with lower and higher transfusion burden, it worked well for the patients who had lower or higher erythropoietin level and for patients with ring sideroblasts. In patients without ring sideroblasts, the rate of transfusion dependence was somewhat similar between luspatercept and ESA, but the duration of response was longer with luspatercept. My opinion is that it is good to have a drug that works across the entire spectrum in the frontline management of patients with lower-risk MDS.
Q: The MEDALIST trial demonstrated that luspatercept has a manageable safety profile without drug-related mortality or increased risk of transformation to acute myeloid leukemia (AML).2 How does the safety in the COMMANDS trial compare with the known safety profile of luspatercept?
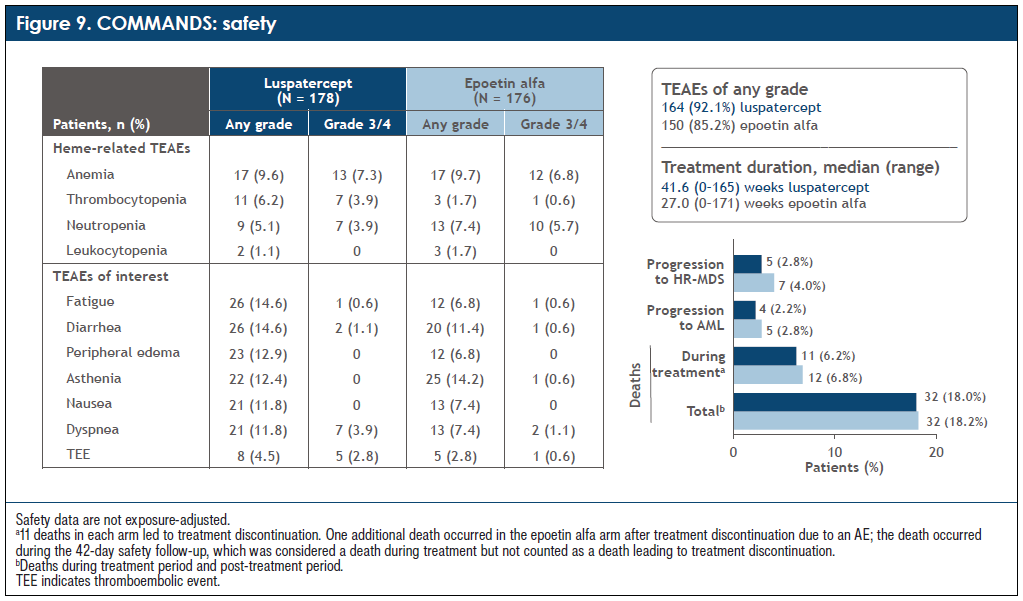
A: The MEDALIST trial did give us a very good sense of the safety profile of luspatercept which led to the initial approval of luspatercept after ESA failure. The main side effect was fatigue, which was generally not severe. The fatigue gets better with continued treatment and there are very low rates of treatment discontinuation due to fatigue. Other side effects we sometimes see are myalgia or muscle pains. Serious side effects are rare and again it’s probably one of our safest drugs to use for patients with LR-MDS. An important item to note is there was no increase in the rate of progression to acute myeloid leukemia, high-risk MDS, or death, so the safety profile is pretty good.
In my opinion, keeping patients on one treatment with a low toxicity profile and a manageable safety profile is important. In the COMMANDS trial, 15.7% of patients receiving luspatercept discontinued treatment compared with 32.4% of patients receiving epoetin alfa due to lack of efficacy (Figure 8). The main side effect for patients was fatigue, which was experienced by 15.2% of patients receiving luspatercept compared to 7.4% of patients receiving epoetin alfa (Figure 9). Fatigue generally improves with continued treatment and tends to not lead to treatment discontinuation, making luspatercept one of the safer drugs to give to patients with LR-MDS. Luspatercept is also easy to dose with a subcutaneous administration once every 3 weeks. All these variables combined translate into clinically meaningful advantages for patients.
Q: How might the COMMANDS data impact treatment decisions and clinical practice for patients with LR-MDS?
A: As luspatercept is already approved after ESA failure for patients with transfusion-dependent anemia with LR-MDS who have ring sideroblasts, and with the COMMANDS data currently under FDA approval, hopefully we may soon see an approval of this drug that will change the first-line management for patients with LR-MDS with anemia. This is the first and only time that we have a drug that is better than ESA in the front-line management for this patient group and, if approved, it could provide an important option leading to high rates of transfusion independence and durability. There are other studies which will examine luspatercept prior to the patient becoming transfusion dependent. This will answer a very important question: Why should we wait until patients are transfusion dependent to treat them when we know anemia can have negative impacts? We should aim to not only improve anemia, but to have a complete resolution, especially when we have good drugs that are active. Also, understanding the sequencing of treatment, for example, the efficacy of ESA after luspatercept or in combination. All these research questions are important next steps in the management of patients with anemia and LR-MDS.
The COMMANDS clinical trial clearly shows that luspatercept is a very effective anemia-correcting drug leading to significant improvement in hemoglobin and transfusion independence among patients who are not previously treated with LR-MDS and are transfusion dependent compared to ESA. The safety profile of luspatercept is good and it’s easy to imagine that luspatercept would lead to a change in the standard-of-care management of patients with LR-MDS.
Dr Zeidan was paid for his contribution to this interview.
References
- Myelodysplastic Syndrome (MDS) research funded by LLS. Leukemia and Lymphoma Society. www.lls.org. Accessed July 24, 2023. https://www.lls.org/research/myelodysplastic-syndrome-mds-research-funded-lls.
- Garcia-Manero G. Myelodysplastic syndromes: 2023 update on diagnosis, risk-stratification, and management. Am J Hematol. 2023;8(8):1307-1325. doi:10.1002/ajh.26984.
- Platzbecker U, Della Porta MG, Santini V, et al. Efficacy and safety of luspatercept versus epoetin alfa in erythropoiesis-stimulating agent-naïve, transfusion-dependent, lower-risk myelodysplastic syndromes (COMMANDS): interim analysis of a phase 3, open-label, randomized controlled trial [published online ahead of print 23 June 9]. Lancet. 2023;S0140-6736(23)00874-7. doi:10.1016/S0140-6736(23)00874-7.
- US FDA accepts Priority Review for Supplemental Biologics License Application (BLA) and EMA validates application for Reblozyl (lustpatercept-aamt) as first-line treatment of anemia in adults with lower-risk myelodysplastic syndroms (MDS). News release. Bristol Myers Squibb. May 1, 2023. Accessed July 24, 2023. https://bwnews.pr/43Y9dOe.
About the Expert
Amer Zeidan, MBBS, MHS, has focused on clinical and translational research in the development of novel therapies for myeloid malignancies, with a special focus on targeted therapies and immunotherapy-based approaches. He is also active in health outcomes and comparative effectiveness research for blood cancers and their therapies and serves as the principal investigator of many investigator-initiated, cooperative group and industry-sponsored clinical trials for myeloid malignancies. He is a member of the MyeloMATCH precision medicine initiative of the National Cancer Institute for both MDS and AML and is very active within the Cancer Therapy Evaluation Program and Early Therapy Clinical Trial Network in working on early-phase clinical trials of novel therapies for myeloid malignancies.
Dr Zeidan has received several prestigious awards, including the Leukemia and Lymphoma Society Scholar in Clinical Research award, the National Cancer Institute Cancer Clinical Investigator Team Leadership award, the AAMDSIF/Evan’s Foundation-MDS Clinical Research Consortium Fellowship award, and multiple other achievement awards. He serves on the editorial board and is a reviewer of several hematology and oncology journals. Dr Zeidan has authored more than 260 peer-reviewed publications and book chapters.